Research
免疫チェックポイント阻害剤の効き目を左右する体内動態要因の解明
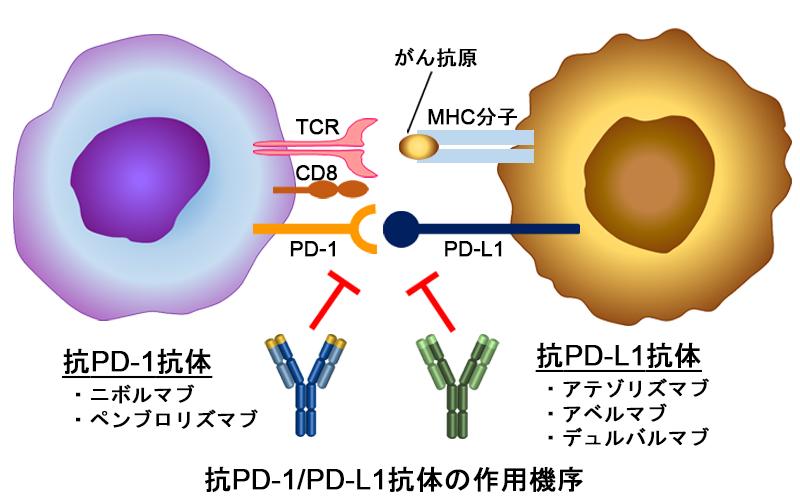
In recent years, cancer immunotherapy such as immune checkpoint inhibitors (ICIs) and CAR-T cells have been approved as new modalities (treatment methods). Cancer immunotherapy has enabled the treatment of cancers that were previously difficult to treat, and has been reported to show dramatic therapeutic effects. On the other hand, their drug efficacy has been observed in only about 20-30% of patients , and the establishment of markers and methods to predict drug efficacy remains to be investigated. In addition, unconventional side effects (immune-related adverse events; irAE) have become a problem in the clinic. ICIs are high-molecular-weight antibody drugs, and the process from administration to drug efficacy and side effects is different from that of conventional low-molecular-weight compounds, but basic research and verification of clinical issues are not necessarily sufficient. We are investigating the relationship between pharmacokinetics and drug efficacy, analysis of drug efficacy mechanism, and elucidation of the mechanism of side effects using about 20 different tumor-bearing mouse models to elucidate the clinical problems of antibody drugs, mainly immune checkpoint inhibitors.